We’ve found that tubed insulin pumps connect a separate device to your body via flexible tubing, while tubeless systems integrate everything into a single adhesive pod worn directly on your skin. Tubed systems offer larger insulin reservoirs and disconnection flexibility, but tubing can snag or kink. Tubeless pumps eliminate these complications and provide greater discretion, though they require complete pod replacement every few days. Both integrate with continuous glucose monitors for automated delivery. The choice between systems depends on your activity level, lifestyle preferences, and comfort priorities—factors we’ll explore throughout this thorough comparison.
Key Takeaways
- Tubed pumps have external reservoirs connected by tubing to infusion sets, while tubeless systems integrate all components into one adhesive body-worn unit.
- Tubeless systems eliminate tubing-related complications like kinking and snagging, but require complete pod replacement versus refillable tubed reservoirs.
- Tubed pumps offer larger insulin capacity and disconnect flexibility, while tubeless provides discretion and continuous delivery during water activities.
- Upfront costs range $5,000-$8,500 for both types, with tubeless averaging $0-$200 monthly supplies versus $0-$300 for tubed systems.
- Both systems integrate with CGMs for automated insulin delivery, though tubeless users report slightly higher satisfaction scores than tubed users.
How Tubed and Tubeless Insulin Pumps Work: Core Design Principles
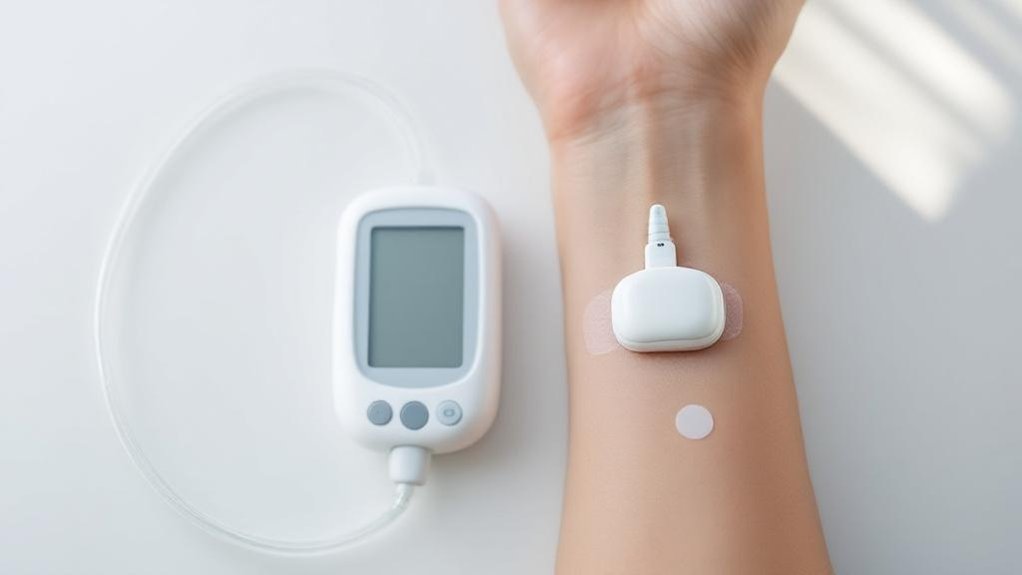
When you’re considering insulin pump therapy, understanding the fundamental design differences between tubed and tubeless systems helps you make an informed choice that aligns with your lifestyle and management preferences.
Tubed pumps separate core pump mechanics into distinct components: a main device housing the insulin reservoir connects via flexible tubing to a remote infusion set. You’ll wear the pump on your belt or in a pocket while tubing delivers insulin to your insertion site.
Tubeless systems integrate everything—reservoir, cannula, and delivery mechanism—into one adhesive unit worn directly on your body. This streamlined approach eliminates external tubing and visible hardware.
Each design offers distinct design advantages. Tubed pumps provide larger reservoir capacity and dedicated on-device controls. Tubeless systems deliver discretion and freedom from tubing-related complications during daily activities. Research indicates that the pump design influences initiation efficiency, with healthcare professionals able to set up patients on therapy in varying timeframes depending on the system architecture.
Daily Life With Each Pump Type: Practical Considerations for Activities and Movement
Your insulin pump becomes part of nearly every movement you make throughout the day, which means the design you choose directly impacts how you navigate physical activities, water exposure, clothing choices, and routine tasks. Tubed pumps typically require disconnection during showers or swimming, creating hyperglycemia risk if reconnection is forgotten—retrospective studies show post-shower reconnection errors occur in 12% of users. They also risk dislodgement during contact sports as tubing snags on objects or shifts during cycling and running. Tubeless pumps offer greater activity flexibility with waterproof designs that maintain continuous insulin delivery during water exposure. Their minimalist profile eliminates tubing entanglement risks during sports, dance, or gym workouts, though extreme heat risks insulin degradation if reservoirs remain exposed beyond thirty minutes. The tubeless design uses one-time use pods that integrate the reservoir and cannula directly into a single disposable unit. Both systems present distinct movement challenges requiring personalized consideration.
Advanced Technology Features: CGM Integration and Automated Delivery Systems
Beyond the physical considerations of pump design, the integration between insulin pumps and continuous glucose monitors represents the most significant technological advancement in diabetes management over the past decade. We’ve seen hybrid closed-loop systems emerge that make automated adjustments to basal insulin based on CGM accuracy and real-time glucose trends. The insulin algorithms differ prominently between tubed and tubeless systems—Medtronic’s Guardian 4 integration allows glucose targets as low as 100 mg/dL, while Omnipod 5 offers broader sensor compatibility with Dexcom and Abbott devices. User feedback consistently highlights the trade-off between proprietary systems and third-party flexibility. As technology improvements continue and future innovations expand sensor options, we’re moving toward more personalized automated delivery that adapts to individual patterns without constant manual intervention. These automated systems help users reach their time in range goals more effectively while reducing the burden of manual insulin calculations.
Safety Profiles and Potential Complications: What Users Need to Know
While both pump types deliver insulin safely when functioning properly, they present distinct risk profiles that deserve your careful consideration. Tubing creates potential points of failure—from accidental disconnections during physical activity to kinking that interrupts delivery without immediate detection. Tubeless systems eliminate these tubing-specific risks, but introduce different concerns around complete device failure and the need for immediate pod replacement to maintain insulin delivery.
In a recent 12-week study involving 104 adults with type 1 diabetes, no severe hypoglycaemia or diabetic ketoacidosis was reported in either the tubeless automated insulin delivery group or the tubeless sensor-augmented pump group, demonstrating favorable safety profiles for both systems.
Tubing-Related Risk Factors
Tubing systems introduce several mechanical vulnerabilities that affect more than half of insulin pump users. Tubing kinking occurs in 64.1% of users, often during sleep or movement, causing hyperglycemia without triggering alarms. Blockage frequency reaches 54.3%, with 4.6%-15% of infusion sets failing within three days. Disconnection events represent the most common precipitating factor for pump failure, leading to diabetic ketoacidosis and requiring hospitalization in 32% of cases. Air bubbles affect 39% of users, disrupting insulin flow and causing unstable glucose patterns. Tubing leakage at connection points increases with extended use and hot weather, resulting in emergency department visits in 32% of significant incidents. Accidental catheter traction during physical activity or clothing changes represents a frequent everyday complication that can interrupt treatment and necessitate infusion set replacement. We recognize these vulnerabilities impact your daily confidence in managing diabetes with traditional pump systems.
Cannula Displacement Considerations
Cannula displacement represents a distinct mechanical failure pattern that affects both tubed and tubeless insulin pump systems. We’ve observed that proper cannula maintenance greatly reduces displacement incidents, particularly when combined with adherence to 48-72 hour replacement schedules. Environmental factors—including heat, humidity, and sweat—compromise adhesive integrity and increase detachment risk. Displacement prevention requires meticulous site preparation, correct insertion technique, and twice-daily visual inspections of insertion sites.
The clinical consequences demand our attention: displacement interrupts insulin delivery immediately, potentially triggering hyperglycemia or diabetic ketoacidosis within hours. We recommend implementing essential safety protocols including mandatory site rotation every two days, immediate replacement following accidental traction, and maintaining backup rapid-acting insulin for emergency use. Understanding these displacement mechanisms empowers us to prevent complications effectively. Patients should evaluate insertion site selection based on their individual profiles, as placement in unhealthy tissue can compromise insulin absorption and contribute to infusion set failure.
Financial Investment: Comparing Upfront and Ongoing Expenses

Understanding the financial commitment of insulin pump therapy requires examining both the substantial initial investment and the continuous costs that follow. Without insurance, upfront costs for new insulin pumps range from $6,000 to $8,000 or more, with tubeless systems like Omnipod starting at $5-$1,260 and tubed systems like Medtronic 780G ranging from $0-$8,574 depending on features and coverage.
Beyond the initial purchase, ongoing expenses add $2,000 to $6,000 annually for essential supplies. We see tubeless systems averaging $0-$200 monthly, while tubed pumps range $0-$300 monthly for infusion sets, reservoirs, and disposable components. Insurance coverage varies greatly—tubeless systems typically fall under pharmacy benefits, while tubed pumps use durable medical equipment channels, creating different affordability pathways that require careful evaluation. Research shows that tubeless automated insulin delivery systems can offset their higher treatment costs through reduced hypoglycemia and ketoacidosis expenses over time.
Physical Comfort and Discretion: Visibility and Wearability Factors
One of the most personal aspects of choosing between insulin pump systems comes down to how the device feels on your body and whether others can see it during daily life.
Pump visibility differs greatly between systems. Tubeless patches adhere directly to your skin with no external tubing, creating a streamlined profile under most clothing. Tubed pumps feature visible tubing connecting the pump unit to your infusion site, though you can minimize this by clipping the pump to clothing or using specialized accessories.
Wear comfort varies based on your lifestyle. Tubeless designs eliminate tubing that might catch on objects or create pressure points during movement. However, tubed systems offer flexibility—you can position the pump unit separately from your infusion site, which some find more comfortable during sleep or physical activity. The Omnipod sensor placement is versatile, allowing you to apply it to various locations on your body just under the skin for optimal comfort.
Maintenance Requirements: Site Changes and Component Replacement Schedules
Whether you choose a tubed or tubeless insulin pump, you’ll need to establish a consistent maintenance routine centered around regular site changes. Both systems require site change frequency of every two to three days to maintain peak insulin absorption through proper site rotation.
The component replacement process differs considerably between systems. With tubed pumps, you’ll separately replace reservoirs, tubing, and infusion sets while your main pump unit lasts for years. You can refill reservoirs from insulin vials when needed. Tubeless pods represent an all-in-one approach—you’ll replace the entire integrated unit containing both reservoir and cannula at each change.
Understanding these maintenance patterns helps you prepare adequately. If failures occur, tubed systems allow component-specific replacement, while pod failures require complete unit replacement and access to your insulin supply. Survey data from 896 respondents with diabetes revealed that tubeless pump users reported higher satisfaction scores compared to those using tubed systems, with ratings of 66.6 versus 64.1 respectively.
Choosing the Right System: Key Advantages and Trade-offs for Different Lifestyles
Your lifestyle shapes which insulin pump system will serve you best. We’ve observed that active individuals and parents often prefer tubeless systems for unrestricted movement during sports, childcare, and water activities. These pumps eliminate tubing that can dislodge during biking or interfere with physical contact, while their discreet profile suits various social settings.
Tubed systems offer flexibility to disconnect during intimate moments or water exposure, though this requires remembering to reconnect. User experiences reveal that lifestyle preferences greatly influence satisfaction—athletes value continuous delivery without disconnection, while others appreciate tubed pumps’ diverse infusion set options. Tubed pump users report no issues even after 20 years of experience, demonstrating the reliability of these systems for long-term management.
Time efficiency also matters. Tubeless initiations require 13 minutes less than tubed systems, and site changes typically demand less commitment than reservoir management, supporting different daily routines.
Frequently Asked Questions
Can I Switch Between Tubed and Tubeless Pumps Mid-Prescription?
Switching requires new prescription adjustments and prior authorization from your insurance, as pump compatibility differs considerably between systems. We recommend coordinating with your healthcare team to guarantee medical necessity documentation supports your change between tubed and tubeless technologies.
Do Airlines Allow Insulin Pumps Through Security Screenings?
Yes, airlines allow insulin pumps through security, but you’ll need to declare your device before screening. Airport regulations permit metal detectors, though manufacturers often prohibit body scanners. We recommend requesting pat-downs instead.
How Do I Manage Pump Alarms in Quiet Settings?
We recommend pump alarm strategies like vibration mode, temporary alert silencing for 30-minute to 24-hour periods, and night-specific profiles. These quiet setting tips help you manage notifications discreetly while maintaining critical safety alerts.
Are Replacement Pods Available at Local Pharmacies for Urgent Needs?
Replacement pod availability varies by location. We recommend calling your local pharmacy ahead, as many major chains carry Omnipod inventory. Your pharmacy options include retail locations, specialty pharmacies, and mail-order services for urgent needs.
Can I Wear Both Pump Types During MRI Scans?
No, neither pump type is MRI-safe. Both tubed and tubeless pumps must be completely removed before scanning due to MRI compatibility concerns and pump safety risks, including potential device damage and dangerous insulin delivery disruptions.
Conclusion
We’ve explored the key differences between tubed and tubeless insulin pump systems, from their core mechanics to daily practicalities. The right choice depends on your unique lifestyle, clinical needs, and personal preferences. We encourage you to discuss these factors with your diabetes care team, trial systems when possible, and connect with other pump users. Your decision should support both ideal glucose management and quality of life. There’s no universally “best” option—only what’s best for you.
